Chemistry
Types of Chemical Reactions
admin
Reactions can be classified on the basis of their specific properties. Since there is no uniform criteria for classification, one reaction can fall into various types simultaneously.
Newland’s Law of Octaves
admin
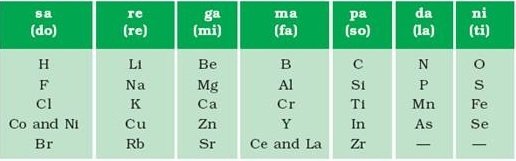
In the year 1864, the British chemist John Newlands attempted the 62 elements known at that time. He arranged them in an ascending order based on their atomic masses and observed that every 8th element had similar properties. On the basis of this observation, Newland’s law of octaves was formulated.
